Electrochemical Reduction of CO2 to Oxalic Acid: Experiments, Process Modeling, and Economics
Electrochemical Reduction of CO2 to Oxalic Acid: Experiments, Process Modeling, and Economics
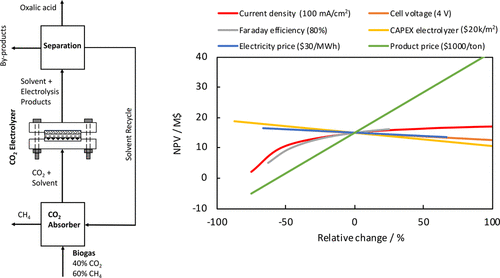
We performed H-cell and flow cell experiments to study the electrochemical reduction of CO2 to oxalic acid (OA) on a lead (Pb) cathode in various nonaqueous solvents. The effects of anolyte, catholyte, supporting electrolyte, temperature, water content, and cathode potential on the Faraday efficiency (FE), current density (CD), and product concentration were investigated. We show that a high FE for OA can be achieved (up to 90%) at a cathode potential of −2.5 V vs Ag/AgCl but at relatively low CDs (10–20 mA/cm2). The FE of OA decreases significantly with increasing water content of the catholyte, which causes byproduct formation (e.g., formate, glycolic acid, and glyoxylic acid). A process design and techno-economic evaluation of the electrochemical conversion of CO2 to OA is presented. The results show that the electrochemical route for OA production can compete with the fossil-fuel based route for the base case scenario (CD of 100 mA/cm2, OA FE of 80%, cell voltage of 4 V, electrolyzer CAPEX of $20000/m2, electricity price of $30/MWh, and OA price of $1000/ton). A sensitivity analysis shows that the market price of OA has a huge influence on the economics. A market price of at least $700/ton is required to have a positive net present value and a payback time of less than 10 years. The performance and economics of the process can be further improved by increasing the CD and FE of OA by using gas diffusion electrodes and eliminating water from the cathode, lowering the cell voltage by increasing the conductivity of the electrolyte solutions, and developing better OA separation methods.