Accelerating CO2 Electroreduction to Multicarbon Products via Synergistic Electric–Thermal Field on Copper Nanoneedles
Electrochemical CO2 reduction is a promising way to mitigate CO2 emissions and close the anthropogenic carbon cycle. Among products from CO2RR, multicarbon chemicals, such as ethylene and ethanol with high energy density, are more valuable. However, the selectivity and reaction rate of C2 production are unsatisfactory due to the sluggish thermodynamics and kinetics of C–C coupling.
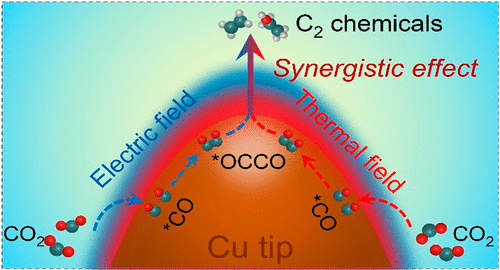
The electric field and thermal field have been studied and utilized to promote catalytic reactions, as they can regulate the thermodynamic and kinetic barriers of reactions. Either raising the potential or heating the electrolyte can enhance C–C coupling, but these come at the cost of increasing side reactions, such as the hydrogen evolution reaction. Here, we present a generic strategy to enhance the local electric field and temperature simultaneously and dramatically improve the electric–thermal synergy desired in electrocatalysis. A conformal coating of ∼5 nm of polytetrafluoroethylene significantly improves the catalytic ability of copper nanoneedles (∼7-fold electric field and ∼40 K temperature enhancement at the tips compared with bare copper nanoneedles experimentally), resulting in an improved C2 Faradaic efficiency of over 86% at a partial current density of more than 250 mA cm–2 and a record-high C2 turnover frequency of 11.5 ± 0.3 s–1 Cu site–1. Combined with its low cost and scalability, the electric–thermal strategy for a state-of-the-art catalyst not only offers new insight into improving activity and selectivity of value-added C2 products as we demonstrated but also inspires advances in efficiency and/or selectivity of other valuable electro-/photocatalysis such as hydrogen evolution, nitrogen reduction, and hydrogen peroxide electrosynthesis.

I value the blog article. Much thanks again. Really Cool. Gregg Ralston
Hello, I wish for too subscribe for this website to obtain most recent updates, therefore where can i do it please assist. Chung Morua
I was examining some of your articles on this internet site and I think this website is really instructive! Retain putting up. Geraldine Miner Fowle
This is the perfect blog for anybody who really wants to understand this topic. Buck Glassel
I am sure this article has touched all the internet people, its really really good article on building up new webpage. Garrett Lapilio
Way cool! Some extremely valid points! I appreciate you penning this write-up and the rest of the site is also very good. Robin Corolla
Currently it looks like Movable Type is the top blogging platform out there right now. Don Holtgrewe
Pretty! This has been an incredibly wonderful article. Thank you for supplying this info. Linwood Berton
Every weekend i used to pay a visit this web page, as i wish for enjoyment, since this this web page conations truly nice funny information too. Norman Mauseth
Hi, I log on to your new stuff on a regular basis. Bernardo Apodaca
Very good article! We are linking to this particularly great post on our website. Wilburn Feenan
This piece of writing provides clear idea designed for the new visitors of blogging, that actually how to do running a blog. Chung Chheng
But wanna remark on few general things, The website design and style is perfect, the subject matter is really fantastic : D. Mason Delmonte
Seeking for an inexpensive high essay creating company to accomplish your papers. We now have top-notch proficient essay writers inside our workforce. Adan Ostrum
Great, thanks for sharing this blog. Really Great. Theodore Moron
We’re a bunch of volunteers and starting a brand new scheme
in our community. Your site provided us with helpful information to work on. You’ve performed an impressive task and our whole community shall be thankful to you.
Visit my web site :: tracfone special coupon 2022
Hello There. I discovered your blog using msn. That is an extremely neatly written article.
I will be sure to bookmark it and return to learn more
of your useful information. Thanks for the post.
I will certainly comeback.
Feel free to surf to my web site – 2022
Muchos Gracias for your post.Really looking forward to read more. Much obliged.
I’d like to thank you for the efforts you have put in writing this
Thanks-a-mundo for the post.Really thank you! Awesome.
My website: порно с дочкой
Awsome site! I am loving it!! Will be back later to read some more. I am bookmarking your feeds also.