Surface hydroxide promotes CO2 electrolysis to ethylene in acidic conditions
Surface hydroxide promotes CO2 electrolysis to ethylene in acidic conditions
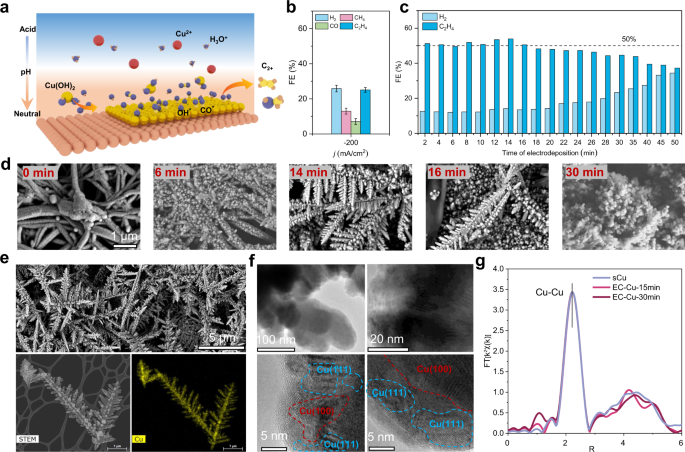
Performing CO2 reduction in acidic conditions enables high single-pass CO2 conversion efficiency. However, a faster kinetics of the hydrogen evolution reaction compared to CO2 reduction limits the selectivity toward multicarbon products. Prior studies have shown that adsorbed hydroxide on the Cu surface promotes CO2 reduction in neutral and alkaline conditions. We posited that limited adsorbed hydroxide species in acidic CO2 reduction could contribute to a low selectivity to multicarbon products. Here we report an electrodeposited Cu catalyst that suppresses hydrogen formation and promotes selective CO2 reduction in acidic conditions. Using in situ time-resolved Raman spectroscopy, we show that a high concentration of CO and OH on the catalyst surface promotes C-C coupling, a finding that we correlate with evidence of increased CO residence time. The optimized electrodeposited Cu catalyst achieves a 60% faradaic efficiency for ethylene and 90% for multicarbon products. When deployed in a slim flow cell, the catalyst attains a 20% energy efficiency to ethylene, and 30% to multicarbon products.

I conceive this site has got some very excellent information for everyone :D. “Believe those who are seeking the truth doubt those who find it.” by Andre Gide.